Unlocking the Molecular Code of Germline Immortality
2026-04-01 11:26
Keywords:lysosomal switch; proteostasis renewal; V-ATPase; C. elegans germ lineage; protein aggregation
Introduction
The perpetuation of life remains one of the most fascinating puzzles in biology: individual organisms are mortal, aging and perishing over time, yet species endure through the immortal nature of the germline. How do germ cells evade the accumulation of damage, maintain their integrity, and faithfully transmit genetic information? A groundbreaking study published in Nature provides unprecedented answers using the model organism Caenorhabditis elegans. The work reveals that germline immortality is not accidental but relies on a specialized molecular switch within the lysosome. Activated at a critical developmental window, this switch boosts lysosomal function, resets proteostasis, and clears cellular damage, safeguarding the health and faithful transmission of germ cells.
1. Protein Aggregation in Oocytes
Using fem-1 mutant female worms that lack sperm, the research team discovered massive accumulation of protein aggregates in proximal oocytes. These aggregates contain various aggregation-prone proteins (e.g., RHO-1, HCP-1, and NMY-2) and manifest as static granules with irreversible fluorescence recovery after photobleaching. These are hallmarks of proteostasis collapse (Fig. 1A-1E). Strikingly, when these females mated with males, sperm signaling triggered the mobilization and clearance of these aggregates within one hour before fertilization (Fig. 1F). This demonstrates that the clearance mechanism is activated by sperm-derived signals, rather than the fertilization event itself.
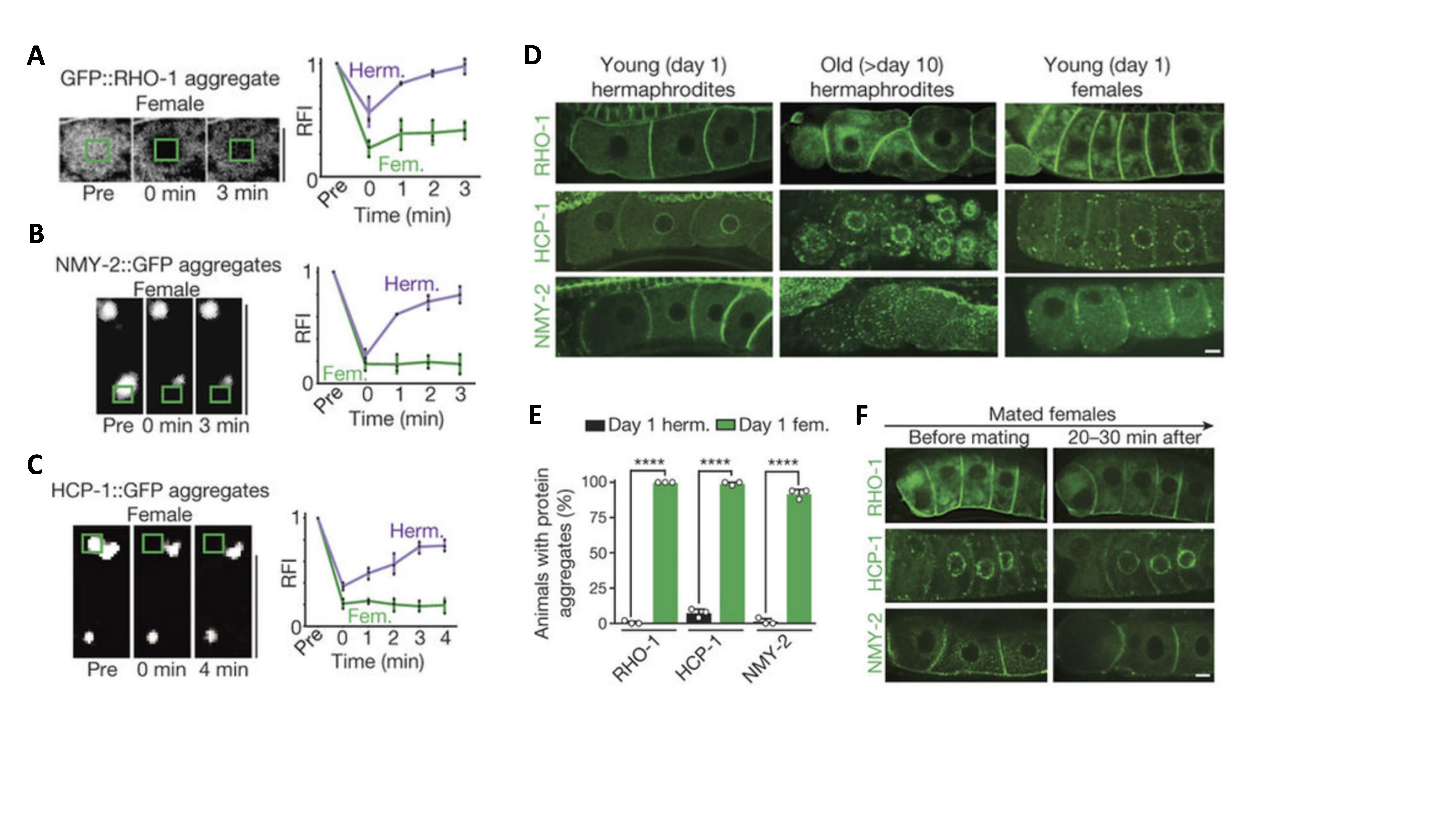
Figure 1. Protein Aggregation in Oocytes
2. A Sperm-Triggered Lysosomal Quality-Control System
Central to this restoration is the activation of vacuolar H⁺-ATPase (V-ATPase), a proton pump that acidifies lysosomes. Knockdown of V-ATPase induced protein aggregation in oocytes (Fig. 2A-2B). Sperm signaling promotes the degradation of GLD-1, a translational repressor that inhibits V-ATPase synthesis, thereby stimulating V-ATPase activity and restoring lysosomal acidification in oocytes (Fig. 2C-2F). Activated lysosomes not only directly promote the engulfment and degradation of protein aggregates but also drive a metabolic switch essential for aggregate mobilization (Fig. 2G-2I).
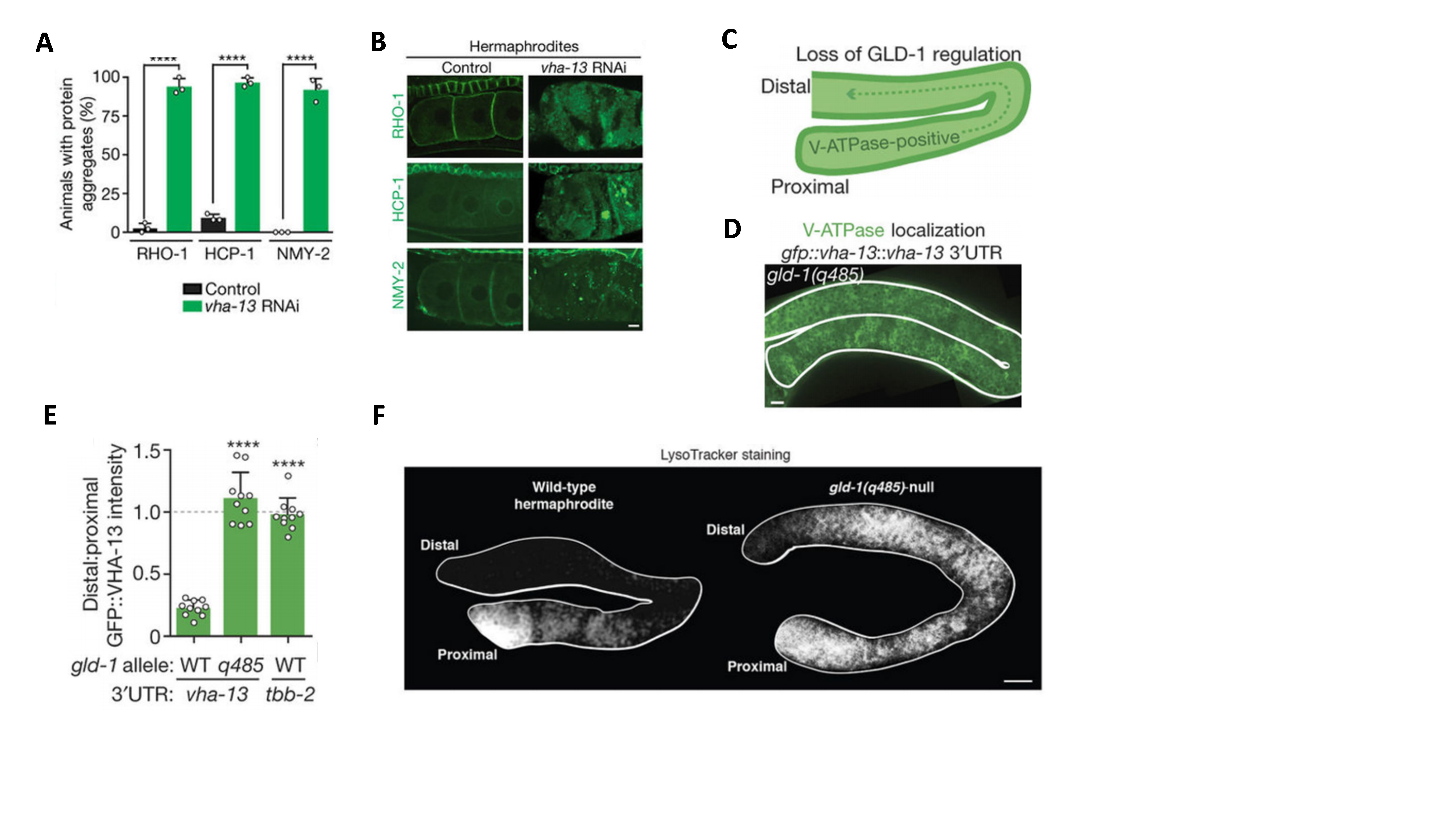
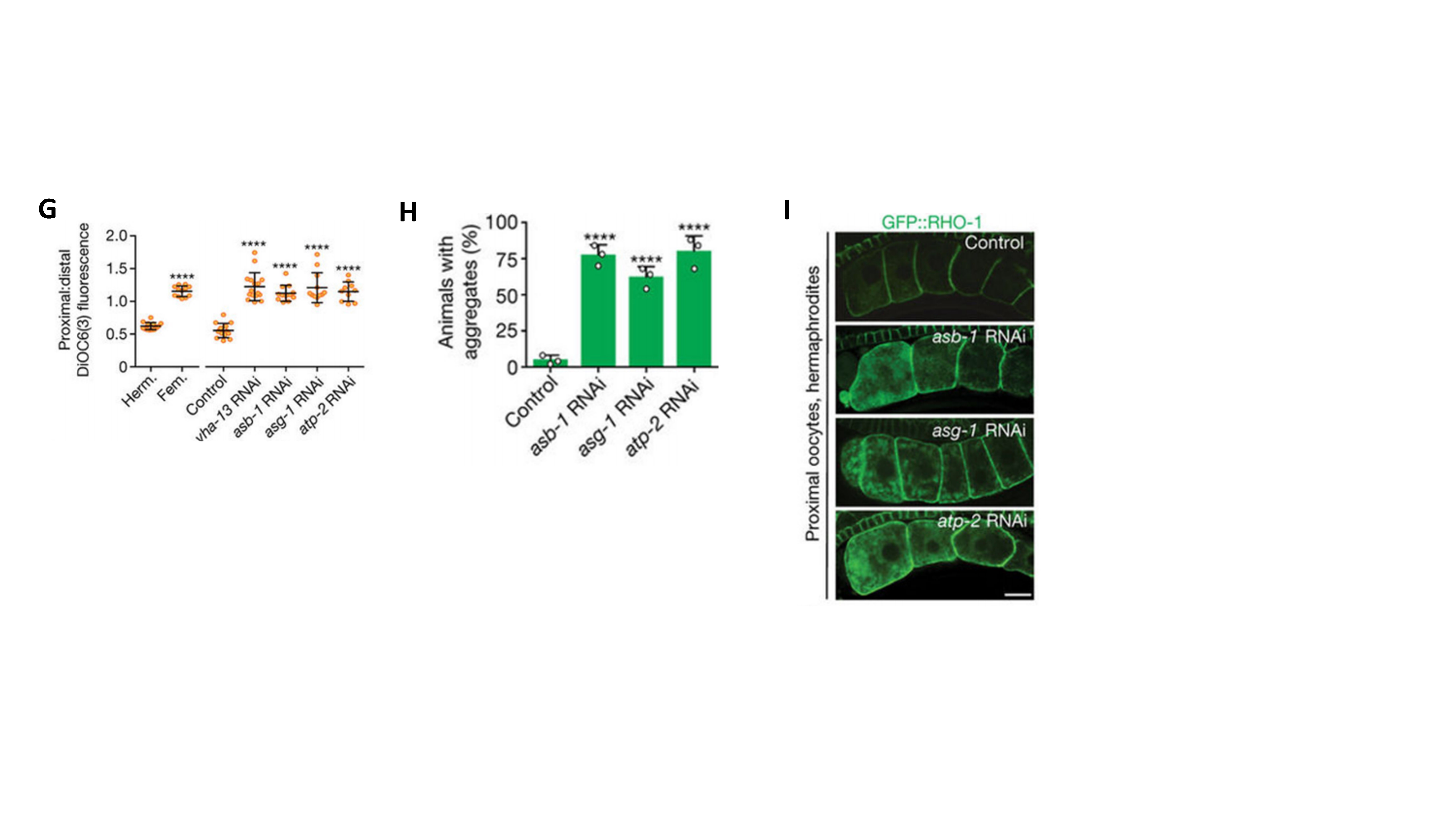
Figure 2. A Sperm-Triggered Lysosomal Quality-Control System
3. Evolutionary Conservation
To test the universality of this mechanism, the researchers validated it in Xenopus oocytes. Lysosomal acidification—marked by increased LysoTracker staining—also occurred during oocyte maturation (Fig.3), suggesting the lysosomal switch is an evolutionarily conserved mechanism that prepares oocytes for fertilization across diverse species.
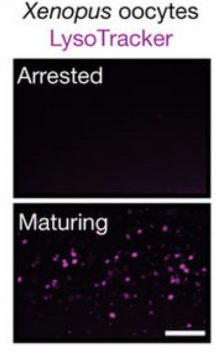
Figure 3. Evolutionary Conservation
Conclusion
This study reveals, for the first time, the central role of a lysosomal switch in ensuring germline immortality: triggered by sperm signaling, this mechanism activates V‑ATPase to enhance lysosomal function, ultimately resetting proteostasis, clearing cellular damage, and safeguarding oocyte and germline integrity. Beyond resolving a long‑standing mystery underlying species perpetuation, this work redefines lysosomes as far more than a mere “cellular waste disposal system,” uncovering their active and essential regulatory functions. It also provides novel targets and strategies for interventions against aging and for the improvement of reproductive health. Although further work is required to fully elucidate the detailed molecular mechanisms, this study establishes a critical foundation for understanding proteostasis and germline homeostasis in fundamental life sciences.
Reference
Bohnert, K. A. & Kenyon, C. (2017). A lysosomal switch triggers proteostasis renewal in the immortal C. elegans germ lineage. Nature, 551(7682), 629–633.
Do you need reliable, custom CRISPR/Cas9 models to drive your own breakthrough research? Contact SunyBiotech (service@sunybiotech.com) today to learn how our C. elegans strain construction services can accelerate your discoveries.






